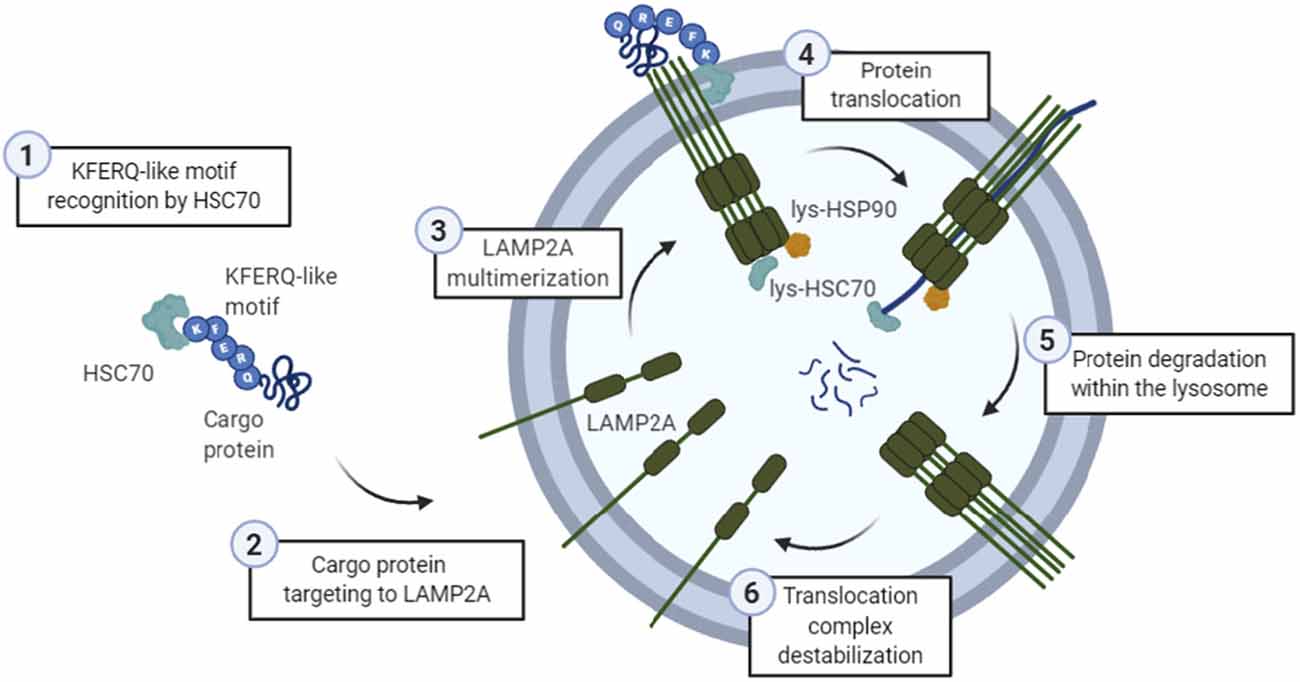
Putative CMA substrates were identified by the presence of a KFERQ-like motif in their sequence ( Dice, 1990) and, using this criterion, it was estimated that ~30% of cytosolic proteins are candidates for CMA ( Wing et al., 1991). After tanslocation, substrate proteins are rapidly degraded by the abundant array of lysosomal hydrolases. Translocation of the substrate across the lysosomal membrane also requires the presence of a luminal form of Hsc70 (lys-Hsc70), which assists in substrate translocation into the lyosomal lumen ( Agarraberes et al., 1997 Cuervo et al., 1997). Internalization of the substrate protein is preceded by its unfolding ( Salvador et al., 2000), a step not required in the other types of autophagy. 
After this targeting step, the substrate protein–chaperone complex docks at the lysosomal membrane through interaction with the cytosolic tail of a single-span membrane protein, the lysosome-associated membrane protein type 2A (LAMP-2A), which acts as a receptor for this autophagic pathway ( Cuervo and Dice, 1996). This motif is recognized by a cytosolic chaperone, the heat shock cognate protein of 70 kDa (Hsc70) that along with its modulatory co-chaperones (Bag1, Hip, Hop and Hsp40) brings the substrate protein to the surface of the lysosomes ( Chiang et al., 1989). We also include a synopsis of the described physiological functions of CMA and some of the connections established between malfunctioning of CMA and disease.įor a protein to be amenable for lysosomal degradation via CMA, the presence of a pentapeptide motif biochemically related to KFERQ in its amino acid sequence is absolutely necessary ( Dice, 1990). In this article and the accompanying poster, we summarize the main steps involved in degradation of cytosolic proteins by CMA, the essential components of this pathway both in the cytosol and at the lysosomal membrane and the basis for the regulation of this autophagic process. This autophagic process, known as chaperone-mediated autophagy, differs from the other forms of autophagy in both the way in which cargo proteins are recognized for lysosomal delivery and the way in which these proteins reach the lysosomal lumen ( Dice, 2007 Cuervo, 2010). A third form of autophagy, solely dedicated to degradation of soluble proteins can also be detected in most cell types in mammals. Lysosomal enzymes can gain access to the enclosed cargo through direct fusion of the vesicles with lysosomes (in macroautophagy), or by internalization of cargo-containing vesicles that form at the lysosomal membrane (in microautophagy). Macroautophagy and microautophagy are variants of the autophagic process, in which entire regions of cytosol (in ‘bulk’ autophagy) or selective cytosolic components (organelles, protein complexes, protein aggregates, pathogens, etc.) are sequestered in vesicular compartments. Various types of autophagy co-exist in almost all cells, and they can be differentiated by the mechanisms that mediate the delivery of cargo (the substrates to be degraded) to lysosomes. The term autophagy (or self-eating) is broadly used to designate the lysosomal delivery and degradation of intracellular components ( Mizushima et al., 2008 Mizushima and Levine, 2010 Yang and Klionsky, 2010). Chaperone-mediated autophagy (CMA) is an intracellular catabolic pathway that mediates the degradation of a selective subset of cytosolic proteins in lysosomes ( Dice, 2007 Cuervo, 2010 Kon and Cuervo, 2010 Orenstein and Cuervo, 2010).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
